Vascular Access: Target Acquired. Anchored in Position. Mission Accomplished.
An overarching goal of Vascular Access Specialists is to expertly provide a patient with a reliable vascular access device that will last until the end of need; for the ‘life of the line’.
In her Vascular Access Talk at the 2022 AVA Scientific Meeting titled “Target Acquired. Anchored in Position. Mission Accomplished.”, Michelle Hawes, RN, DNP, CRNI, VA-BC, ACRP-CP provides expert insight around Vascular Access Securement Device Options, their respective Safety & Efficacy and the elements that clinicians should record when evaluating the best choice for catheter securement. Here we briefly explore the progression from insertion to end of need and highlight points for consideration when evaluating the best option for your patient.

Place the Right CVAD in the Optimal Position
Getting it right. The optimal position for a Central Venous Access Device (CVAD) is the lower 1/3 of the Superior Vena Cava (SVC) or Cavatorial Junction (CAJ); anything less is considered suboptimal and increases the risk for complications. Equally important is keeping the optimally positioned CVAD in place for the life of line; the duration of a patients need for that specific device.
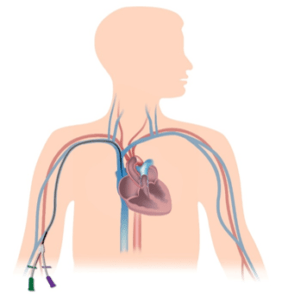
Secure in Position
Interestingly, the time and money that is expended by clinical facilities on CVAD insertion far outweighs that designated to CVAD securement following insertion. There are however securement solutions in practice to keep CVAD’s in their optimal position.
Engineered Securement Devices
- Adhesive Securement Device (ASD)
- Integrated Securement Device (ISD)
- Subcutaneous Anchor Securement System (SASS)
- Tissue Adhesive
Improvised Securement Devices. These are off label and are not recommended due to lack of measured standardization.
- Suture Based Securement (SBS)
- “Creative” Taping
Minimize Complications
While Each Securement Option has its advantages and disadvantages, all are aimed to prevent complications caused by catheter movement such as pistoning, retraction, migration and dislodgement. Securement issues that can impact CVAD patients include Insertion Site Issues, CLABSI, Thrombus and MARSI.

When selecting a securement option, minimizing device migration and dislodgement is an important consideration as is ease of care & maintenance. When paired, these considerations contribute to the success of providing the patient with the one vascular device that will serve them until the end of need.
Simplify Care & Maintenance
In Michelle (Mickey) Hawes’s presentation, she features the Care & Maintenance advantages of SecurAcath®, a Subcutaneous Anchor Securement System (SASS).
- Provision for 360⁰ cleaning while the catheter remains anchored
- Remains with the catheter until the end of need
- No need for repeated device removal and reapplication
- Does not depend on the contracted pharmacy choice of securement (homecare)
- Saves time during routine dressing changes
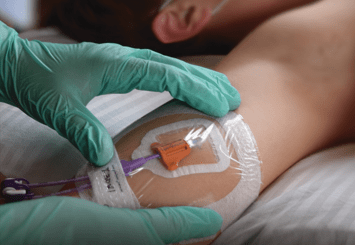
But beyond simplified care & maintenance, Subcutaneous Anchor Securement Systems (SASS), like SecuraCath®, have been repeatedly identified as a superior securement option based on clinical evidence and data.
Mounting Clinical Evidence for Subcutaneous Anchor Securement Systems (SecurAcath®)
A growing body of research shows clear benefits of SASS compared to other securement methods. When selecting a securement option, it’s important to consider its safety and efficacy; The probable benefit should be weighed against any probable injury or illness from using the device.
- In a Systematic Review of the Safety and Efficacy of Central Vascular Access Device Securement conducted by Jon Bell et al, CVAD securement safety and efficacy outcomes are explored. According to 39 data-rich clinical studies, there are clear safety and efficacy benefits presented by a Subcutaneous Anchor Securement System (SASS) like SecuraCath® that are superior to other methods.
- Results from a study conducted by Mark Rowe et al Catheter securement impact on PICC-related CLABSI: A university hospital perspective, showed a 288% increase in relative risk of CLABSI found in the group with an Adhesive Securement Device (ASD) compared to the group being treated with a Subcutanous Anchor Securement System (SASS).
- A landmark study was recently presented at the 2022 World Congress on Vascular Access (WoCoVA) in Athens, Greece. The study compared outcomes for migration or dislodgement and reaching the end of catheter need for patients with two different types of securement – an Adhesive Securement Device (ASD) and a Subcutaneous Anchor Securement System (SASS). The volume of data accumulated over 10 years demonstrated that SASS is the optimal securement method when the goal is to reach the end of need with the original inserted treatment device.
Eloquest Healthcare is committed to providing solutions that can help improve catheter securement and decrease complications while reducing unnecessary costs. For more information about SecurAcath®, please call 1‐877‐433‐7626 or complete this form.
Additionally, we invite you to learn more about facilitating a complimentary Vascular Access Device Site Assessment designed determine the state of vascular access device & dressing securement for patients in your facility. You and your team will be provided with actionable information around your facility’s infection risk, nurse efficiency, and potential product waste. Click here to request a Vascular Access Device Site Assessment today!









