
More Research Needed on Central Vascular Access Device (CVAD) Securement
CVADs are a fundamental part of patient care and are widely indicated and used across many different medical specialties. Due to their prevalence, best practices have been established for CVAD placement. However, surprisingly little research has been done to investigate the effectiveness of securement methods following device placement.1
Targeted research is needed to evaluate the safety and efficacy of different securement methods.1

Comparing Different Securement Methods
There are 3 categories of securement1:

Sutures are a type of transdermal securement commonly used with CVADs. However, sutures have never been classified as an external device securement tool by the FDA. Using SBS off-label for CVAD securement exposes both patients and providers to potential safety concerns, such as catheter dislodgment and needlesticks.1


Many effective options for catheter securement are available. There is substantial evidence that engineered stabilization devices (ESDs), such as ASDs or SASS, are noninferior to SBS. Additionally, the safety and efficacy of ESDs have been studied extensively and demonstrated.1
Ideal CVAD securement should1:
- Be safe for providers
- Improve patient quality of care
- Prevent CVAD line malposition or movement
Systematic Review of Securement Method Safety and Efficacy
Bell, et al. conducted a systematic review of CVAD securement literature from January 2021 to January 2022 to assess reported safety and efficacy outcomes. While there are thousands of published CVAD studies, many do not explicitly address securement methods. However, in studies with direct comparative data on rates of catheter migration and dislodgment, clear benefits for SASS were found.1
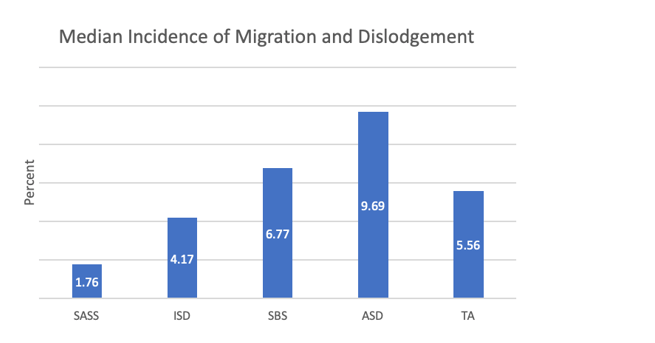
SASS= subcutaneous anchored securement system, ISD= integrated securement device, SBS= suture-based
securement, ASD= adhesive securement device, TA= tissue adhesive.
Figure created with data from Bell, et al. 2022.
However, more research is needed to determine the impact of different securement methods on central line-associated bloodstream infection (CLABSI) and other CVAD complications, as well as the resulting effect on patient outcomes.1
Access the full study here.
Eloquest Healthcare is committed to providing solutions that improve patient quality of care. SecurAcath is a subcutaneous catheter securement system that stabilizes the catheter beneath the insertion site throughout the entire dwell time. To learn more about SecurAcath, visit eloquesthealthcare.com/securacath/ or complete this form to request an evaluation.

References:
- Bell JA, Hawes M, Diloreto E, Gibson SM. Systematic Review of the Safety and Efficacy of Central Vascular Access Device Securement. Journal of the Association for Vascular Access1 September 2022; 27 (3): 15–35. doi: https://doi.org/10.2309/JAVA-D-22-00013.







