
A “STICKY” Situation: Randomized Controlled Trial Investigates Use of Liquid Adhesive to Increase Dressing Adherence & Reduce CVAD Failure
CVADs - Essential Devices with High Failure Rates
Central venous access devices (CVADs) are essential to the timely and effective treatment of critically ill patients. They deliver lifesaving drugs, nutrition, and blood products. Yet despite their importance, these devices frequently suffer from complications, with a reported failure rate of up to 30 percent, often due to inadequate dressing and securement of the catheter.
Due to their location in the neck, internal jugular (IJ) CVADs have a particularly high risk of failure and failure-related complications. Dressings do not adhere well to this area, because of beard growth, clammy skin associated with critical illness, or the ‘drag’ of the multiple infusion lines attached to the CVAD. Such dressing disruptions lead to increases in local and systemic infection – including central line-associated bloodstream infections (CLABSI) and medical adhesive-related skin injury (MARSI) – which adversely affects patient outcomes.
Lack of Research on Dressing & Securement Options
Despite the importance of CVADs to the care of patients, the dressing and securement of these devices has received surprisingly little attention. Only recently has there been a growing realization about the important role that dressing and securement methods play in avoiding catheter-related complications
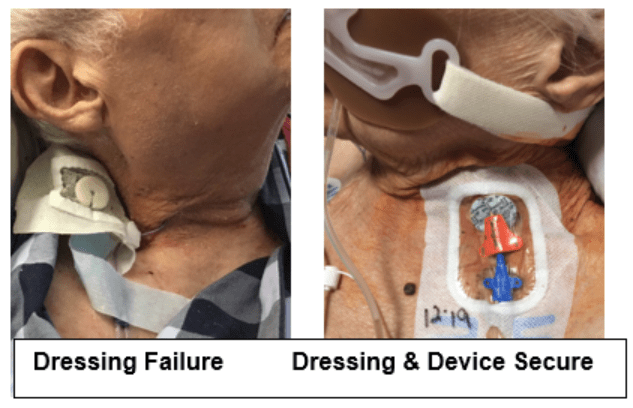
Prior research has shown that the dressings on CVADs placed in the internal jugular (IJ) typically fail within 1-1.5 days, primarily because the adhesive no longer sticks strongly enough. Given that a CVAD may be in place for 7-14 days, this dressing failure is a significant problem that needs to be addressed.
“The fact is that many of these dressings fail, leaving the CVAD poorly secured and exposed to contamination, which adversely affects patient outcomes,” said Professor Nicole Marsh of the Royal Brisbane and Women’s Hospital, Queensland, Australia. “Our research team is committed to improving the care patients receive for their CVADs through innovative dressing and securement strategies.”
The STICKY Trial
To identify possible improvements, Marsh and her colleagues are conducting a randomized controlled trial across four intensive care units to directly compare securement methods. The trial is designed to test whether or not the application of Mastisol® Liquid Adhesive (MLA) can improve internal jugular vein dressing adherence and lead to better CVAD care for patients.

The study is the first randomized controlled trial that aims to assess the clinical and cost effectiveness of MLA in reducing CVAD dressing failure compared to “standard” dressing care. According to Marsh, 160 patients will be recruited for the study.
“Our hypothesis is that the CVAD dressings secured with Mastisol will remain intact more often than those with no Mastisol, thereby protecting the insertion site from external contaminants in addition to securing the catheter in place to prevent movement and dislodgement,” said Marsh. She added that a reduction in CVAD dressing failure will also reduce:
1) The number of potentially painful dressing changes
2) Adverse events, such as infection, dislodgement, and ultimate CVAD failure
3) Clinician workload and cost associated with CVAD failure.
In addition to assessing dressing adherence and CVAD failure, this study will also provide crucial data about outcomes like infection and skin injury, which are of particular importance to patients. While the true incidence of MARSI in adult ICU patients is not clear, it has been reported to occur in 10-22 percent of patients. In this trial, Marsh and her colleagues are performing daily checks of the CVAD insertion site to observe for signs of skin injury and MARSI, which should further quantify the burden of these complications in this patient population.
Marsh said they expect to publish results in mid-2023, which will “contribute much needed evidence on strategies to reduce CVAD dressing failure in critically ill patients.” A summary of the final study results will be provided here following its publication, so be sure to subscribe to the Eloquest Blog or check back frequently to ensure you receive the update.
Eloquest Healthcare is committed to providing solutions that can both improve patient outcomes and reduce unnecessary costs. We can work with your institution to perform a Vascular Access Device Site Assessment, which evaluates vascular access device sites and dressing securement for patients. The assessment measures infection risks, nurse efficiency, and potential product waste, among other important parameters. Click here to request a Vascular Access Device Site Assessment today!








