Frequent Dressing Disruptions
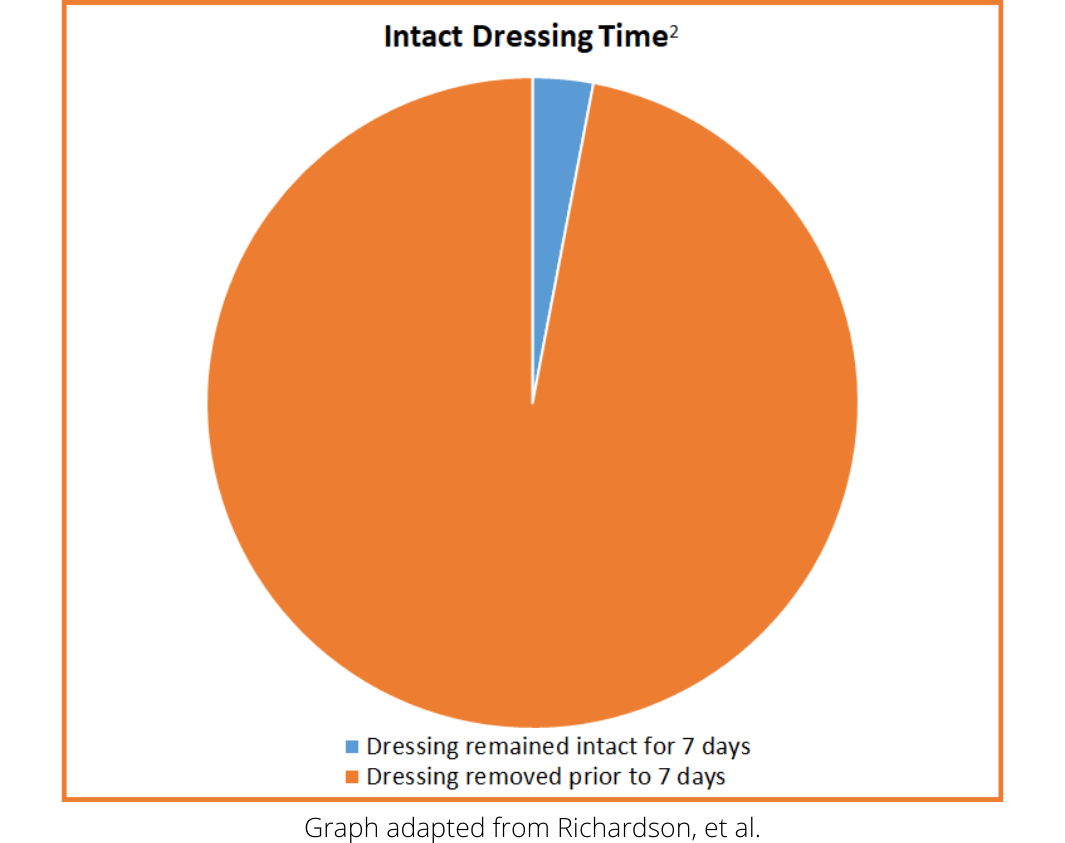
Vascular access dressing disruptions occur frequently during vascular access device (VAD) maintenance, leading to premature dressing changes. This issue has been examined by multiple studies. According to a study undertaken by Timsit, et al., approximately 67% of central venous catheter (CVC) dressing changes are performed before the planned date.1 This can be due in part to the limited durability of common, commercially available CVC dressings. Richardson, et al. found that 75% of evaluated CVC dressings lasted less than 48 hours, with only 3% of dressings maintaining functionality for a full 7 days.2
This dressing fallibility is further seen in a study conducted by Chan, et al., in which the average time between device insertion and the first dressing change ranged between 0.94 to 1.83 days.3 This is a significantly shorter lifespan than the up to 7 days recommended for transparent dressings in the 2021 Infusion Therapy Standards.4 It is concerning that only 3% of evaluated CVC dressings reached the best practice benchmark of up to 7 days intact, as seen in the image to the right.
Catheter-related bloodstream infection (CRBSI) development is a complication seen in vascular access patients, with dressing changes contributing to increased risk of CRBSI occurrence. Each dressing change further increases this risk, with the possibility CRBSI rising by more than 3-fold after the second dressing disruption and more than 10-fold after the final dressing disruption.1
The High Cost of Dressing Disruptions: Patients’ Wellbeing and Clinical Resources
Dressing disruption is not only bad for the patient, but also for the bottom line in facilities. Premature dressing changes are associated with significantly increased:
- Risk of CRBSI development
- Supply costs
- Nursing time
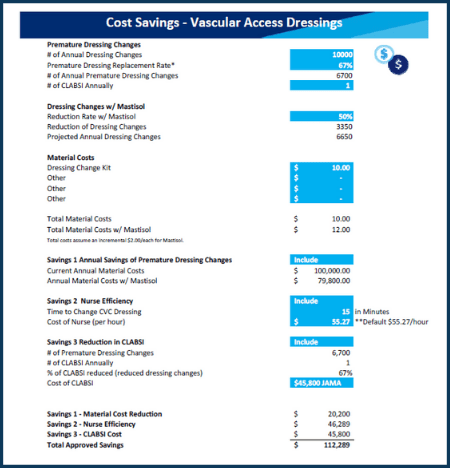
The following table is a cost breakdown of the negative financial impacts associated with dressing disruption.
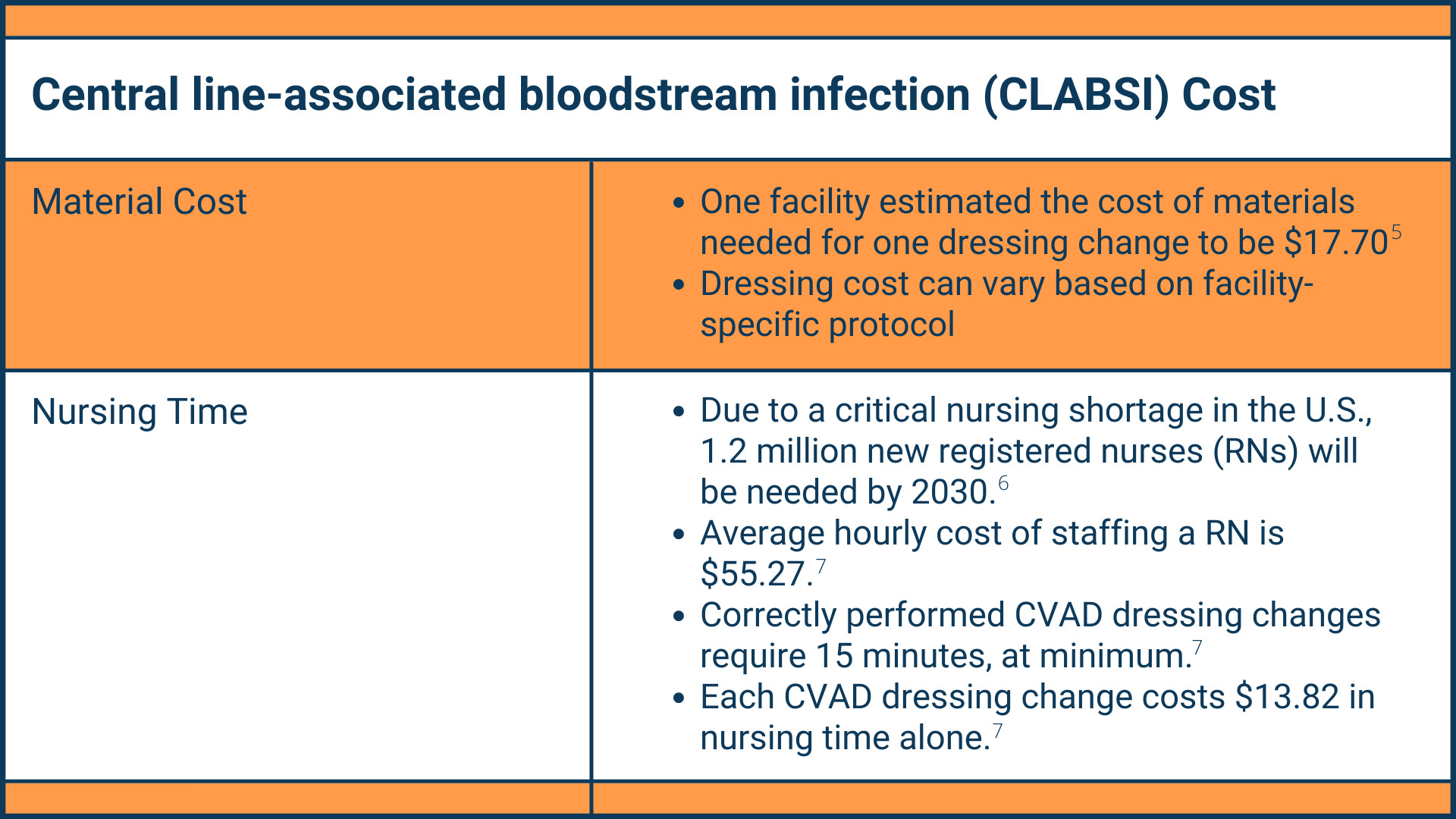
CLABSI cost can be up to $46,6858
Mastisol® Liquid Adhesive: An Effective, Cost-Sensitive Solution
The addition of Mastisol Liquid Adhesive to dressing application and maintenance routines can decrease unnecessary dressing changes. With less dressing disruption, both supplies and nursing time previously spent on premature dressing changes can be better allocated.
Implementation of Mastisol has been shown to increase facility efficiency and patient satisfaction
Over an 8-month period of Mastisol and tissue adhesive implementation, PRN dressing changes decreased by 31% and loose dressing prevalence decreased by 16%. The addition of Mastisol and tissue adhesive to dressing routines decreased overall cost and risk of patient harm. Based on results obtained in the 8-month timeframe, a cost savings of approximately $40,000 per year was estimated.5
The cost of an extra CVC dressing is double the expense of Mastisol, not including staff time
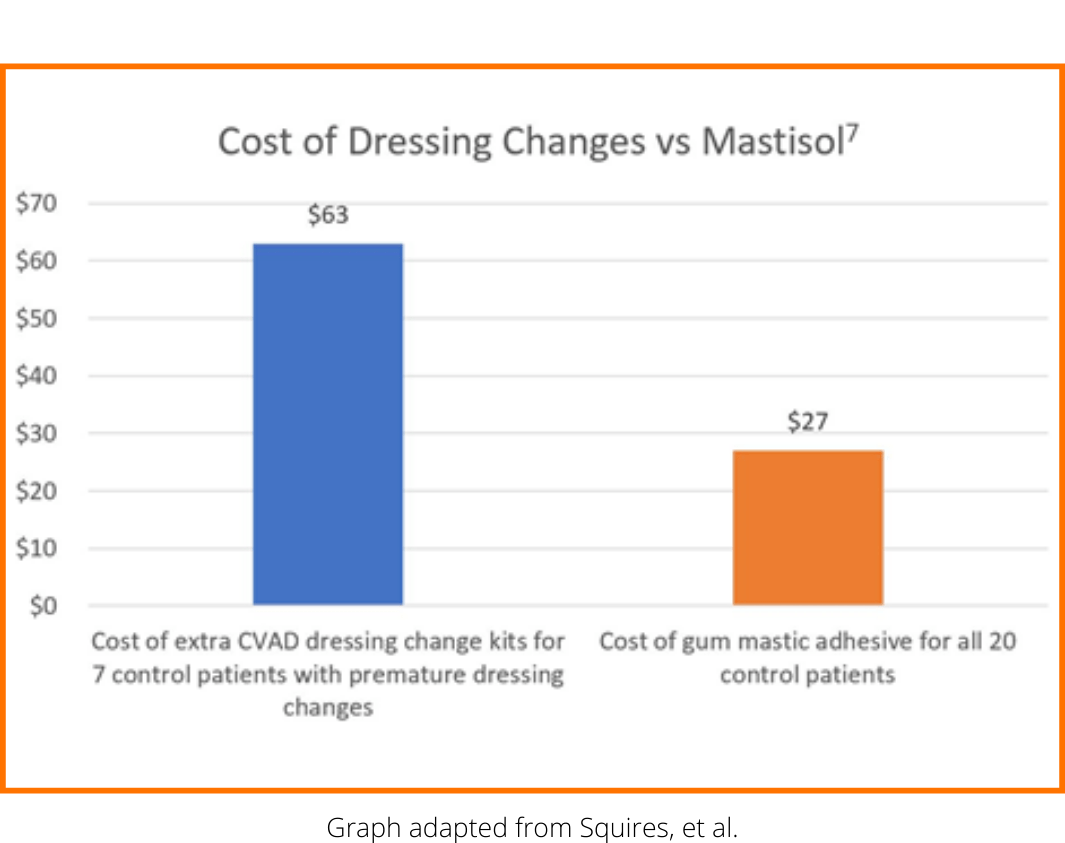
Another study was done to evaluate Mastisol’s effectiveness in reducing dressing disruption and corresponding CLABSI rates. Among the 33 patients who received central venous access device (CVAD) site care with Mastisol application, 0 premature dressing disruptions were reported. In the control group of 20 patients treated without Mastisol, early dressing disruption occurred in 7 patients at a prevalence of 35%. No CLABSI development was observed in either patient group. It is important to note that the cost of the additional CVAD dressings needed for 7 patients is double the expense of Mastisol for all 20 control patients, excluding staff time.7
In one study, Mastisol use was also associated with lower CLABSI rates in central line dressing sites, reducing occurrence to 0 over 514 line days.9 Reducing CLABSI rates is not only beneficial for patient outcomes, but financially impactful as well. Preventing one case of CLABSI a year would result in over $30,000 saved, as well as covering the cost of Mastisol and Detachol for one unit.9
Free Downloadable Cost Calculator
To calculate the potential cost that could be avoided at your facility, please download our free Cost Savings Calculator. Input the number of annual dressing changes and known material costs at your facility to get an estimation of such costs. Contact your local representative for help in performing the analysis or for help in collecting observational data on your facility’s prevalence of non-intact dressings.
Eloquest Healthcare is committed to providing solutions that can help increase dressing adherence and decrease risk of skin injury, while reducing unnecessary costs. For more information about Mastisol Liquid Adhesive, please call 1‐877‐433‐7626 or complete this form.

References
- Timsit J, et al. Dressing disruption is a major risk factor for catheter-related infections. Crit Care Med. 2012; 40(6): 1707-1714.
- Richardson A, et al. Central venous catheter dressing durability: an evaluation. Journal of Infection Prevention. 2015; 16(6): 256–261
- Chan RJ, Northfield S, Larsen E, et al. Central venous Access device Securement And Dressing Effectiveness for peripherally inserted central catheters in adult acute hospital patients (CASCADE): a pilot randomised controlled trial. Trials. 2017;18(1):458. doi:10.1186/s13063-017-2207-x.
- Infusion Therapy Standards of Practice: Vascular Access Device Assessment, Care, and Dressing Changes. J Infus Nurs. 2021;44:S119-S123
- Jameson L, From PRN to Routine… A Vascular Access Team’s Journey to Reducing PRN Central Venous Catheter Dressing Changes By Adding to the Vascular Access Tool Box. Presented at: Association for Vascular Access Scientific Meeting, September 15-18, 2018; Columbus, OH.
- The 2021 American Nursing Shortage: A Data Study. University of St. Augustine for Health Sciences blog. May 2021. Available at: https://www.usa.edu/blog/nursing-shortage/.
- Squires EN, Bruggeman S. Use of gum mastic medical adhesive to maintain central venous access device dressing integrity. Presented at: Association for Vascular Access Scientific Meeting, October 4-7, 2019; Las Vegas, NV.
- Nelson RE, Angelovic AW, Nelson SD, Gleed JR, Drews FA. An economic analysis of adherence engineering to improve use of best practices during central line maintenance procedures. Infect Control Hosp Epidemiol. 2015 May;36(5):550-6.
- Bortz A, Hardinger K, Peltzer J. Impact of implementing Mastisol in central line dressing changes on central line-associated blood stream infections in progressive care units: a quality improvement project. Presented at: Magnetizing KC Symposium, February 24, 2015.